In the game, users are presented with a chemical equation, and they are tasked with adjusting the number of atoms of each element in the equation to balance it. The simulation provides a virtual environment where users can interact with atoms and molecules to learn how to balance chemical equations. “Balancing Chemical Equations” is designed to help users learn how to balance chemical equations. The Museum Science Chicago chemical reaction game: Hence, here are a few games that can be played as a classroom exercise or even at home! 1. With many chemistry games available online, kids can also engage in a few balancing chemical equation games that can help them become more friendly with the concept. Grab your lab coat and safety goggles, and let’s dive into the world of chemical reactions and online games! Making understanding balancing chemical equations facile through online games 
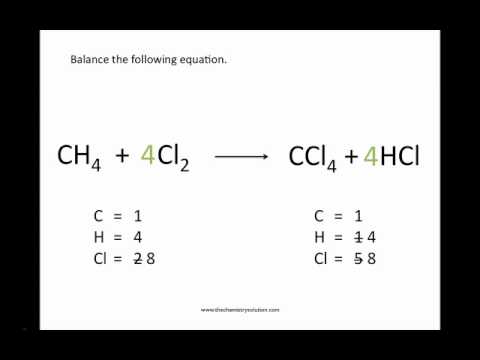
So, get ready to ignite your chemistry skills with these exclusive online games that will challenge your mind and help you master the art of balancing chemical equations. In this case, online games can serve as a fun and interactive way to help students learn and practice balancing chemical equations. Whether you’re a student trying to ace your next test or a curious learner looking to deepen your understanding of chemistry, fun activities, and online interactive games are the ideal way to make learning about chemical reactions fun and engaging. However, it can be difficult to grasp and memorize the rules and methods for balancing chemical equations. It is an essential step in predicting the outcome of a chemical reaction and understanding the stoichiometry of the reaction. Remember that if there's no coefficient in front of an element, it's assumed that the coefficient is 1.Balancing chemical equations is a fundamental concept in chemistry that helps to understand the behavior of different chemicals and how they react with each other. Now the number of atoms in each element is the same on both sides of the equation, so the equation is balanced. To balance this, add the coefficient 2 before H2 on the left side of the equation so there are 4 hydrogen atoms on each side, like 2H2 + O2 → 2H2O. However, subscripts can't be changed and are always multiplied by the coefficient, which means there are now 4 hydrogen atoms on the right side of the equation and only 2 hydrogen atoms on the left side. For the equation H2 + O2 → H2O, you would add the coefficient 2 before H2O on the right side so that there are 2 oxygen atoms on each side of the equation, like H2 + O2 → 2H2O. To balance the equation, you'll need to add coefficients to change the number of atoms on one side to match the other. Since the number of atoms in each element isn't identical on both sides, the equation is not balanced. There are 2 hydrogen atoms and 1 oxygen atom on the right, so you would write "H=2" and "O=1" under the right side. 
For the equation H2 + O2 → H2O, there are 2 hydrogen atoms being added to 2 oxygen atoms on the left, so you would write "H=2" and "O=2" under the left side. For example, your equation should look something like "H2 + O2 → H2O." Count the number of atoms in each element on each side of the equation and list them under that side. To balance a chemical equation, first write out your given formula with the reactants on the left of the arrow and the products on the right. If the value you assigned returns coefficients that have a greatest common factor (GCF), simplify the chemical equation by dividing each value by the GCF.If there is only one fraction, multiply all values by that values denominator. If the value you assigned returns fractional values, just multiply all values by the least common multiple (LCM) of the denominators to get rid of the fractions.This shows us the values are as follows:.Since H: 2b = 3c + d, we can calculate b like this:.Then start solving the system of equations to get the following values: To quickly do this, take one variable and assign a value to it.You must find the one where every variable is in its smallest, non-fractional form. Since there are more variables than equations, there are multiple solutions. Solve this system of equations to get the numeric value for all the coefficients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
